Cervical cancer is the fourth most common malignancy diagnosed in women and causes 270’000 deaths every year1. In 2008, the Nobel Prize of medicine was awarded to Dr. Herald zur Hausan – a German virologist and medical doctor – for his work and discoveries on HPV causing cervical cancer2. His discoveries led to a worldwide joint effort by virologists, epidemiologists, molecular biologists and clinicians to tackle the disease by preventing viral infection through the development of efficient prophylactic vaccines for HPV3. Vaccines are highly efficient and prevent the majority of cancer. However, cancer prevalence is still high and developing efficient therapies is of extreme importance. In this article, we will discuss epidemiological aspects of cervical cancer, the virology of HPV and how zebrafish are valuable assets in the field.
Cervical cancer epidemiology
Cervical cancer is the 4th most common cause of cancer in women after breast, colorectal and lung cancer4,5. 70% of cervical cancers are squamous cell carcinomas and develop in the epithelial tissue lining the outer part of the cervix6,7. The remaining cases are adenocarcinomas and develop in glandular cells of the cervix. Adenocarcinomas are harder to diagnose as they occur higher up in the cervix6. All women are at risk of developing cervical cancer and the risk increases after 30 years of age.
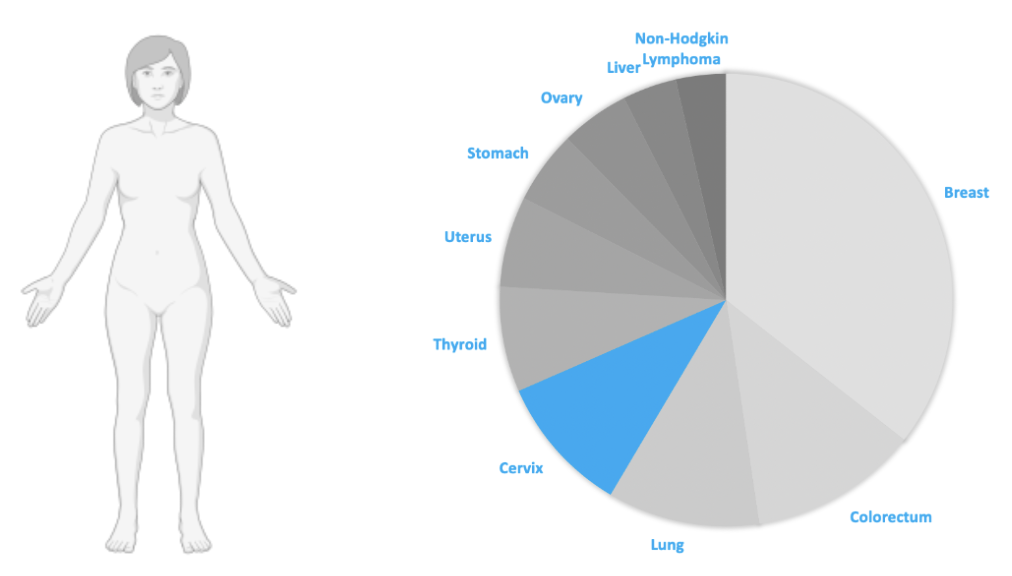
99% of cervical cancers develop as a result of a persistent infection with a human papillomavirus (HPV)5,6. HPV is the most common sexually transmitted infection and one of the most common viral infections worldwide. Most sexually active men and women will be infected at least once in their lives but will not necessarily develop any related pathology8,9. Indeed, most HPV infections are harmless and will clear spontaneously. HPVs are classified as high or low risk and are associated with pathologies ranging from malignant cancers to cutaneous warts9. High-risk HPV infections in particular account for 70% of cervical cancers and pre-cancer lesions and therefore represent a strong epidemiologic risk factor which is at the heart of the fight against cervical cancer.
Human Papillomavirus Infection
HPVs are small non-enveloped double stranded DNA viruses which are about 50-60 nm in diameter. The viral genome encodes for 8 functional and 2 structural proteins which enable viral replication and assembly respectively9.
HPVs only infect undifferentiated epithelial cells in the basal layer of the epithelium which have a high prolific activity but only generate new infectious viral particles once the cell has fully matured and has been pushed to the outermost layer of the tissue. To reach and infect the target cells, the virus depends on microlesions and abrasions of the epithelial surface. Following infectious internalisation, viral DNA is released in the cell cytoplasm and transported to the nucleus where gene expression and DNA amplification is tightly regulated by the infected epithelial cell. To insure viral replication in mature and senescent epithelial cells which do not possess a functional replicative machinery, HPVs encode two specific proteins which promote cell proliferation, perpetuate cell cycle and inhibit cellular apoptosis, altogether enabling viral DNA replication, viral protein synthesis and assembly into new viral particles which are then shed at the epithelial surface3.
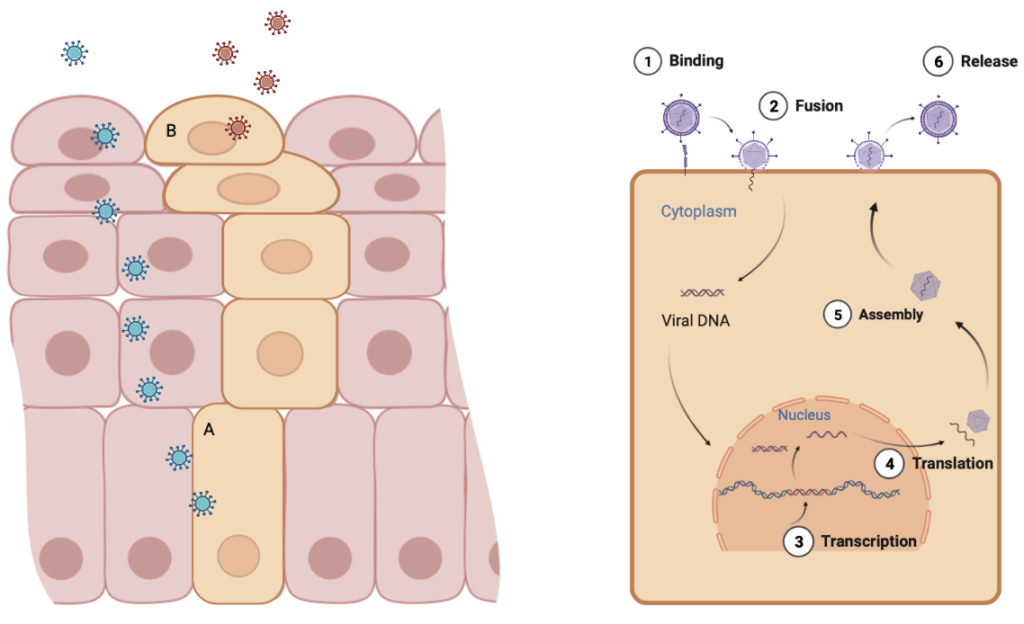
In the course of one HPV infection cycle, thousands of HPV genomes are created and released by a single infected epithelial cell. However, the principle issue does not reside in the number of shed virions but rather in the prolonged cell cycle and increased proliferation induced by the virus. Indeed, by encoding functional replicative proteins, HPV inhibits all of the negative cell-cycle regulators and control checkpoints present in healthy cells which prevent cells from accumulating genetic mutations. Infected cells therefore replicate endlessly and accumulate countless genetic alterations, eventually causing malignant cell transformation into invasive cancer cells. High-risk HPV strains encode very effective functional proteins which interact with several tumour-suppressor genes, promoting malignant transformations and cancer development.
HPV prevention and treatment
HPV is an important and global epidemiological burden with severe consequences. In August 2020, the World Health Organisation (WHO) adopted the Global Strategy for cervical cancer elimination which aims at decreasing cervical cancer incidence rate below 4 cases/100’000 women and eliminating the disease by 203010. The approach relies on 3 main pillars which include prevention strategies as well as efficient treatments. Vaccination and annual screening stand respectively at the first and second line of defence against HPV infection and cancer development. As of today, three effective prophylactic vaccines are available and target a broad spectrum of high and low-risk HPV strains, providing near to 100% protection against viral infection and disease development11.
Screening
Vaccination
Treatments
Vaccines were developed to prevent viral infection only and therefore do not clear pre-existing lesions. Despite the availability of prophylactic vaccines, infection rates and cancer prevalence remain high, supporting the need for efficacious treatment strategies. While most HPV infections clear spontaneously, the process is slow and does not exclude future relapse and disease development. In that sense, immunotherapies in the form of therapeutic vaccines could boost cell-mediated immune responses, accelerate infection clearing and decrease morbidity and mortality associated to HPV infections.
Cancer treatment development and zebrafish assays
Preventing and treating HPV infection is essential to lower the incidence of cervical cancer in women. However, when HPV bypasses the immune system and infects the cervical epithelium, viral lesions can become malignant and develop into cancer. Development of cancer treatments requires acute comprehension of the disease pathogenesis. Over the past years, zebrafish assays brought valuable insight into cancer development and metastatic dynamics as well as potential treatment targets, altogether contributing to progress in cervical cancer treatment strategies.
Zebrafish are robust research models for cancer research, mainly due to the evolutionary conservation of cancer related programs between zebrafish and humans and due to their transparent development enabling in-vivo tumour growth visualisation. Indeed, zebrafish xenotransplant assays enable live visualisation of tumour formation, progression and migration. Fluorescently labelled tumour cells are injected in zebrafish embryos which are visualised with different microscopy techniques during the following days to observe tumour growth and development.
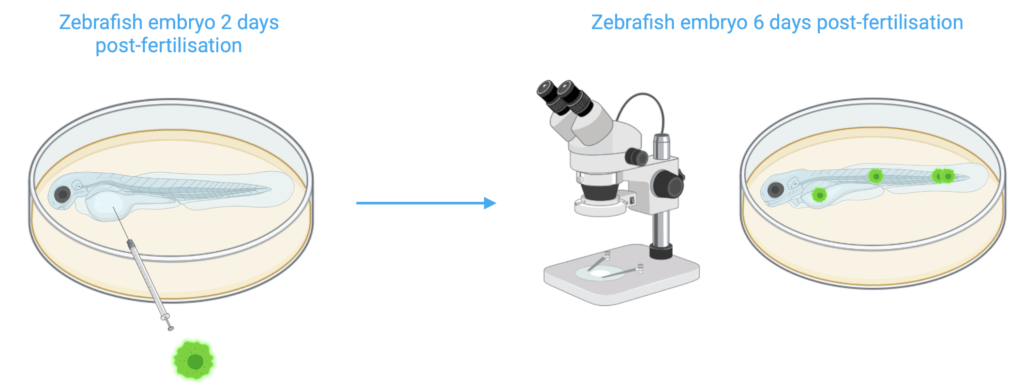
Multiple papers report using zebrafish tumour models for the discovery of key pathways leading to cancer development and metastatic progression12–14. Additionally, zebrafish models have been constructed for angiogenesis and toxicity visual analysis in the development of various drug therapies. Recently, anti-cervical cancer effects of a novel prodrug were investigated in zebrafish tumour models15. Prodrugs are compounds which are only converted to pharmacologically active drugs through the metabolism and therefore hold less adverse events than other traditional cancer treatments. Zebrafish proved to be a good system to evaluate the toxicity, accurate targeting and prodrug effect on tumour invasion and metastasis, confirming efficiency for cervical cancer therapy.
Although prophylactic vaccines and annual screening policies have decreased cervical cancer prevalence, HPV infection rates and cervical cancer remain a global health issue. Developing efficient treatments is thus essential and zebrafish have proven to be excellent assets in this field.
References
- Small, W. et al. Cervical cancer: A global health crisis: Cervical Cancer: A Global Health Crisis. Cancer 123, 2404–2412 (2017).
- The Nobel Prize in Physiology or Medicine 2008. NobelPrize.orghttps://www.nobelprize.org/prizes/medicine/2008/hausen/facts/.
- Crosbie, E. J., Einstein, M. H., Franceschi, S. & Kitchener, H. C. Human papillomavirus and cervical cancer. The Lancet 382, 889–899 (2013).
- Buskwofie, A., David-West, G. & Clare, C. A. A Review of Cervical Cancer: Incidence and Disparities. J. Natl. Med. Assoc. 112, 229–232 (2020).
- Cervical cancer. https://www.who.int/westernpacific/health-topics/cervical-cancer.
- Basic Information About Cervical Cancer | CDC. https://www.cdc.gov/cancer/cervical/basic_info/index.htm (2021).
- Definition of cervical squamous cell carcinoma – NCI Dictionary of Cancer Terms – National Cancer Institute. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/cervical-squamous-cell-carcinoma (2011).
- Human papillomavirus (HPV) and cervical cancer. https://www.who.int/news-room/fact-sheets/detail/human-papillomavirus-(hpv)-and-cervical-cancer.
- Kombe Kombe, A. J. et al. Epidemiology and Burden of Human Papillomavirus and Related Diseases, Molecular Pathogenesis, and Vaccine Evaluation. Front. Public Health 8, 552028 (2021).
- Cervical Cancer Elimination Initiative. https://www.who.int/initiatives/cervical-cancer-elimination-initiative.
- Harper, D. M. & DeMars, L. R. HPV vaccines – A review of the first decade. Gynecol. Oncol. 146, 196–204 (2017).
- Castro-Oropeza, R. et al. Adipose-derived mesenchymal stem cells promote the malignant phenotype of cervical cancer. Sci. Rep. 10, 14205 (2020).
- Zhang, Y. et al. CircCDKN2B-AS1 interacts with IMP3 to stabilize hexokinase 2 mRNA and facilitate cervical squamous cell carcinoma aerobic glycolysis progression. J. Exp. Clin. Cancer Res. 39, 281 (2020).
- Kim, H.-K. et al. TMBIM6/BI-1 contributes to cancer progression through assembly with mTORC2 and AKT activation. Nat. Commun. 11, 4012 (2020).
- Chen, H.-C. et al. Evaluation of the anti-cervical cancer effect of a prodrug :CBZ-AAN-DOX with hypoxic cell culture and tumor-bearing zebrafish models. Exp. Cell Res. 391, 111980 (2020).
