Water pollution is evolving into a serious human and environmental health problem. Numerous pollutants can be found in waters, including synthetic compounds, chemicals naturally present in the environment and endocrine disruptors. To ensure good water quality and safe consumption, drinking water must be certified free of these toxins. An effective and accurate quality control system is therefore essential. Over the past few years, zebrafish have made their way into this field and can now be used as in-vivo water quality control systems. To find out more, read on!
Environmental health
As the world’s population grows and global demands expand, technological advances and industrial activities are constantly increasing. Evolving manufacturing processes allow for the development of new synthetic compounds that add to the burden of natural chemicals in the environment, resulting in the exposure of humans and wildlife to toxic substances. Indeed, the environment holds numerous toxic compounds including metals, arsenic, lead and endocrine disruptors which have been found to have profound effects on human or animal health1. There is therefore an urgent need to identify the consequences of toxin exposure, to understand the biological, chemical and genetic mechanisms underlying the effects of toxin exposure, and to develop effective ways of assessing the levels of natural and chemical toxins in the environment (including water, soil and air).
Bellow some common water contaminants and their adverse health effects2–5:

Arsenic
Arsenic is a metalloid which occurs naturally in rocks and may enter drinking water through natural deposits or runoffs from agriculture. Arsenic can have serious adverse effects in children and adults, including stomach pain, vomiting and impaired nerve function. Long-term exposure over several years due to contaminated water can even lead to cancer.

Cadmium
Cadmium is a metal and environmental pollutant derived from industrial or agricultural sources and which can enter drinking water sources through soils, due to human activity or corrosion of pipes and well components. Cadmium exposure is thought to be linked to several cancers including breast, lung, kidney and pancreas among others. Data also suggests that cadmium may be a risk factor for osteoporosis.

Bisphenol A
BPA is a chemical compound widely used in the plastic industry. It is known as an endocrine disruptor as it is a xenoestrogen, meaning that it mimics oestrogen hormone properties. BPA can be found in industrial food and beverage packaging and has therefore important health implications. BPA can be detrimental for children’s brains and prostate glands and may also affect their behaviour.

Nitrates
Nitrates are chemical compounds naturally present in the environment, particularly in water and soil. Increasing concentrations of nitrates have been observed in water as a result of anthropogenic pollution, mainly of agricultural origin. Ingestion of nitrates in drinking water for instance has be linked to several adverse health effects, including colorectal, bladder and breast cancer as well as methemoglobinemia in babies.
Environmental health is an essential field of research for both humans and wildlife as it attempts to answer fundamental questions related to toxin exposure which can ultimately lead to the development of preventive measures to protect and improve both human and animal health. It is therefore crucial to have a reliable method to understand the effects of low dose, cumulative exposures or exposures to several toxicants, what biological processes can be affected by toxicant exposure, if the effects can be observed in subsequent generations or if there are methods to treat toxicant exposure and to reverse undesirable effects7.
The zebrafish biosensor
To address these questions, researchers have turned to zebrafish. Indeed, zebrafish are valuable research models and are at the forefront of toxicology research7,8. Their short generation time and transparent development make them an ideal tool to study long-term effects of toxicant exposure. In-vivo observation of toxic effects provides an ideal tool to evaluate toxicity and measure toxicant absorption, distribution, metabolism and elimination. Additionally, the important number of offspring generated at each breeding enable longitudinal studies of toxicant effects at a population scale.
In the context of environmental toxicology, zebrafish are commonly used for biomonitoring. Transgenic zebrafish lines have been developed to express fluorescent proteins in the presence of a chemical or environmental toxin of interest, providing a system to monitor levels of toxic compounds in the environment7. For instance, a transgenic zebrafish line has been engineered to express GFP (Green Fluorescent Protein) in the liver after exposure to oestrogen, providing a specific and sensitive in vivo test system to facilitate and accelerate detection of oestrogenic compounds in aquatic environments9.
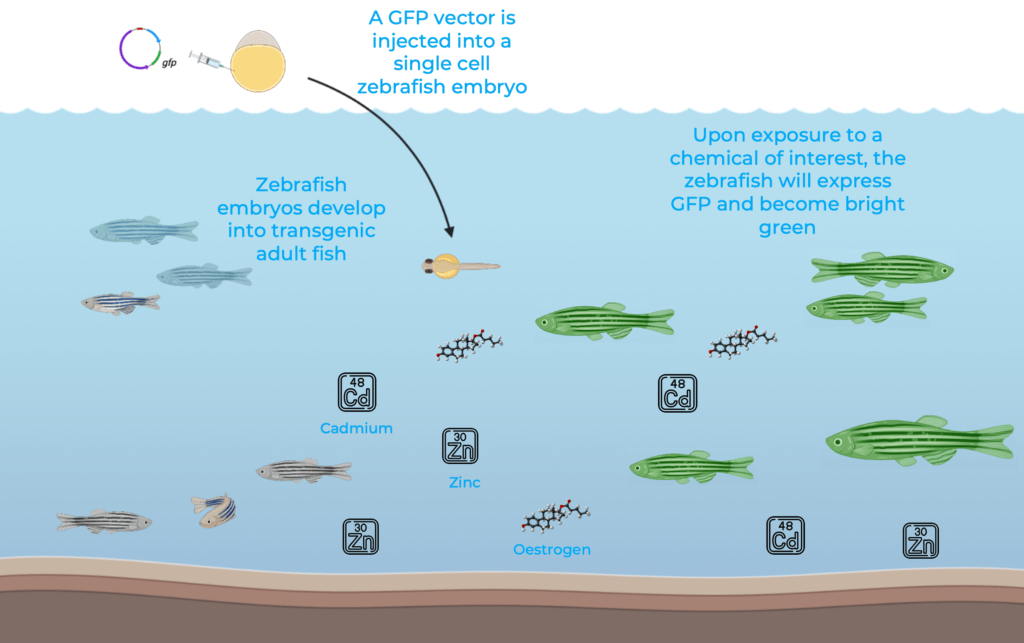
Similarly, transgenic zebrafish were generated to assess aquatic levels of zinc and cadmium, two heavy metals and important water pollutants. By coupling GFP to a metal-sensitive gene vector that was injected into single-cell zebrafish embryos, scientists developed a transgenic zebrafish line sensitive to zinc and cadmium in a dose and time-dependent manner10. Transgenic zebrafish have become valuable biosensors to detect toxic levels of heavy metals, endocrine disruptors, chemicals and other organic pollutants and are therefore important tools for water quality control.
The Zebrafish Embryo Acute Toxic Test
The fish acute toxicity test is the most frequently used method for chemical hazard identification, environmental risk as well as water quality assessments. Fish are exposed to a sample for a period of 96 hours and abnormalities and fish lethality are recorded at 24, 48, 72 and 96 hours. The lethal concentration (LC50) of the sample is determined by the concentration which kills 50% of the tested fish11. This test is a core requirement for safety assessments by many global regulatory frameworks for manufactured chemicals. Yet, it raises several economic and ethical concerns . Indeed, the test requires a large number of fish that undergo significant suffering as the experiment ends in animal death12. Over the past years, the method has been refined to reduce the number of fish required but the mortality endpoint could not be advanced to an earlier endpoint11.
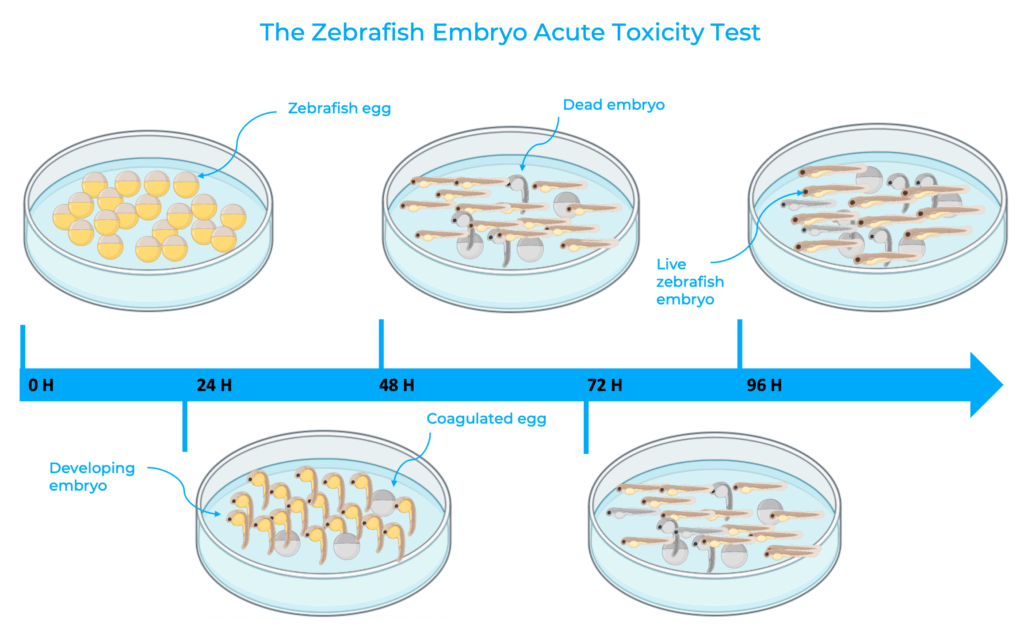
The Zebrafish Embryo Acute Toxicity Test (ZFET) is an ethical and economic alternative to the fish acute toxicity test. This method is based on zebrafish eggs and assesses viability of zebrafish embryos, therefore no longer exposing live and healthy fish to toxicants8,13. Similarly to the fish acute toxicity test, the ZFET test exposes the zebrafish eggs to a sample for 96 hours and determines the LC50 using the concentration that was lethal to 50% of the embryos. The ZFET test is therefore a sustainable solution to reduce the number of juvenile and adult fish used for aquatic toxicity testing and water quality controls.
In conclusion, zebrafish are proving to be an excellent tool for environmental toxicity studies and water quality testing, while reducing the number of animals used in experiments, lowering costs and improving output data. In our increasingly polluted world, zebrafish seem to have a promising future.
References
- Bambino, K. & Chu, J. Zebrafish in Toxicology and Environmental Health. in Current Topics in Developmental Biology vol. 124 331–367 (Elsevier, 2017).
- Arsenic in Drinking Water | HealthLink BC. https://www.healthlinkbc.ca/healthlinkbc-files/arsenic-drinking-water.
- Tips to reduce BPA exposure. Mayo Clinic https://www.mayoclinic.org/healthy-lifestyle/nutrition-and-healthy-eating/expert-answers/bpa/faq-20058331.
- Genchi, G., Sinicropi, M. S., Lauria, G., Carocci, A. & Catalano, A. The Effects of Cadmium Toxicity. Int. J. Environ. Res. Public. Health 17, 3782 (2020).
- Ward, M. et al. Drinking Water Nitrate and Human Health: An Updated Review. Int. J. Environ. Res. Public. Health 15, 1557 (2018).
- US EPA, O. Basic Information about Lead in Drinking Water. https://www.epa.gov/ground-water-and-drinking-water/basic-information-about-lead-drinking-water (2016).
- Shao, Y. et al. Integrated zebrafish-based tests as an investigation strategy for water quality assessment. Water Res. 150, 252–260 (2019).
- von Hellfeld, R., Brotzmann, K., Baumann, L., Strecker, R. & Braunbeck, T. Adverse effects in the fish embryo acute toxicity (FET) test: a catalogue of unspecific morphological changes versus more specific effects in zebrafish (Danio rerio) embryos. Environ. Sci. Eur. 32, 122 (2020).
- Chen, H. et al. Generation of a fluorescent transgenic zebrafish for detection of environmental estrogens. Aquat. Toxicol. 96, 53–61 (2010).
- Liu, L., Yan, Y., Wang, J., Wu, W. & Xu, L. Generation of mt:egfp transgenic zebrafish biosensor for the detection of aquatic zinc and cadmium: A biosensor for detecting heavy metals. Environ. Toxicol. Chem. 35, 2066–2073 (2016).
- Katsiadaki, I. et al. Dying for change: A roadmap to refine the fish acute toxicity test after 40 years of applying a lethal endpoint. Ecotoxicol. Environ. Saf. 223, 112585 (2021).
- Burden, N. et al. Key Opportunities to Replace, Reduce, and Refine Regulatory Fish Acute Toxicity Tests. Environ. Toxicol. Chem. 39, 2076–2089 (2020).
- Acute aquatic toxicity: Zebrafish Embryo Acute Toxicity Test (ZFET). https://joint-research-centre.ec.europa.eu/eu-reference-laboratory-alternatives-animal-testing/alternative-methods-toxicity-testing/validated-test-methods/aquatic-toxicity/acute-aquatic-toxicity-zebrafish-embryo-acute-toxicity-test-zfet_en.
