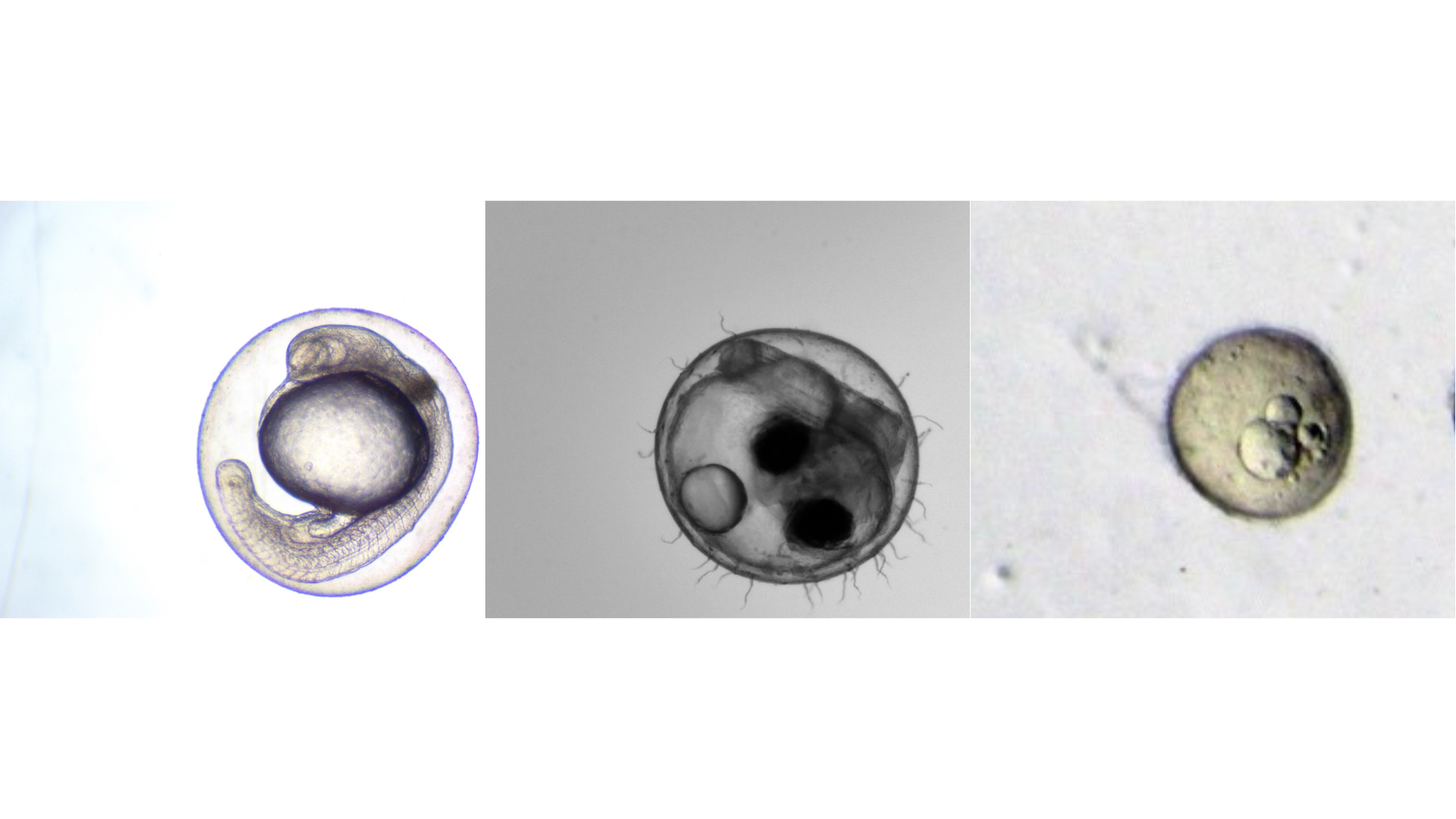
When it comes to toxicology, fish embryo offer something rare: models that are fast, ethically sound, and biologically relevant. But as research scales up, manual workflows can become a bottleneck, limiting both speed and reproducibility. In this month’s article, we explore how automation is changing the game, helping labs unlock the full potential of fish embryo studies for faster, more reliable results.
Introduction
Over the past century, the field of toxicology has expanded dramatically: from drug safety to environmental pollution, the challenges we face are more complex than ever. With growing demands for ethical, high-throughput solutions, researchers need models that deliver both relevance and efficiency. So what makes a model truly fit for modern toxicology?
Why Fish Embryo Models?
- Biologically relevant: Each species brings distinct strengths to toxicology research, making them complementary models for diverse scientific questions. Zebrafish, with over 70% genetic similarity to humans, are widely used for translational research (1). Medaka, known for their temperature resilience and high sensitivity to environmental toxins, excel in ecotoxicological studies. Killifish, with their rapid life cycle, extreme stress tolerance, and diapause capability, are particularly valuable for aging research. A shared advantage of these models is their transparent embryos, which enable real-time observation of developmental and physiological processes
- Rapid and cost-effective: These models develop quickly (e.g., zebrafish hatch in 48-72 hours) and require minimal resources, making them ideal for large-scale, high-throughput studies.
- Ethical and practical: External fertilization allows non-invasive studies from the earliest stages, reducing ethical concerns. Their small size and ease of breeding further support high-throughput screening and automation.
Key Applications and Tests in Toxicology
Fish embryo models are transforming toxicology research by offering efficient, scalable, and biologically relevant alternatives to traditional mammalian testing. Their applications span drug safety, environmental toxicant assessment, and developmental/reproductive toxicity studies, supported by standardized protocols.
Drug safety and efficacy screening
Toxic side effects remain a major challenge in drug development, with many promising compounds failing due to safety concerns. Traditional mammalian toxicity testing, while informative, is often costly, time-consuming, and limited by reproducibility issues. As modern toxicology shifts toward understanding mechanisms of toxicity and reducing animal use, fish embryo models have emerged as powerful alternatives. Zebrafish, in particular, are at the forefront of preclinical drug safety testing, helping to identify toxic compounds early and reduce costly failures in clinical trials. Their high genetic similarity to humans, rapid development, and transparency enable high-throughput screening for cardiotoxicity, hepatotoxicity, and neurotoxicity, key endpoints in drug discovery (2).
Environmental toxicant assessment
Rapid industrialization and chemical production have introduced thousands of pollutants, including synthetic compounds, metals, pesticides, PFAS, and endocrine disruptors, into aquatic ecosystems. Monitoring their impact is critical, as waterborne contaminants can disrupt fish reproduction, development, and survival, with cascading effects on ecosystems and human health. Fish embryo models provide controlled, high-resolution tools to assess early life-stage toxicity and ecological risk.
- Zebrafish are widely used for acute and developmental toxicity screening (3). Their application is supported by standardized assays such as OECD TG 236 (Fish Embryo Acute Toxicity, FET) and ISO 12890:1999 (Embryo & Early Larval Toxicity).
- Medaka embryos are sensitive to metals and other environmental stressors. They exhibit concentration-dependent mortality and developmental deformities under exposures such as selenium and cadmium, supporting life-stage specific risk assessment (4).
- Killifish are emerging ecotoxicology models due to their short life cycle and drought-resistant embryos. Copper exposure studies demonstrate chronic effects including reduced growth, delayed maturation, postponed reproduction, and increased mortality, making them suitable for full life-cycle toxicity testing (5).
Together, these models enable investigation of low-dose exposure, cumulative toxicity, latent developmental effects, and transgenerational impacts, questions that are difficult to address directly in human populations.
Developmental and reproductive toxicity studies
Chemical exposure during early development can disrupt organ formation, endocrine signaling, and reproductive function, with effects that may persist into adulthood or even future generations. Detecting these subtle yet significant impacts requires sensitive, standardized biological models.
Fish embryo and life-cycle assays provide validated frameworks for this purpose.
- Zebrafish are widely used to detect teratogenic, cardiotoxic, neurodevelopmental, and endocrine effects. OECD TG 236 (FET) supports early developmental screening, while OECD TG 234 (FSDT) evaluates sex differentiation and endocrine disruption. Complementary assays such as ZEDTA enable detailed assessment of organ-specific developmental abnormalities.
- Medaka are particularly suited for reproductive and endocrine studies, with well-characterized sex differentiation pathways and validated reporter lines. Early endocrine activity can be assessed using OECD TG 251 (REACTIV) for estrogenic effects and OECD TG 252 (RADAR) for androgen pathway disruption, while longer-term reproductive impacts are evaluated through OECD TG 229 (Fish Short-Term Reproduction Assay) and OECD TG 240 (Medaka Extended One-Generation Reproduction Test), which assess fertility and generational effects.
Together, these models enable scalable assessment of developmental disruption, endocrine activity, fertility impairment, and transgenerational effects, making them powerful tools for environmental and regulatory toxicology.
The Power of Automation
Despite the robustness of fish embryo models, early-stage workflows often rely on manual handling. Embryo screening, sorting, and plating are typically performed by trained operators, making these steps labor-intensive and inherently variable. Subtle differences in developmental stage, morphology, or viability can influence assay outcomes, affecting reproducibility and comparability across experiments, laboratories, and time.
These early sources of variability can propagate throughout toxicity studies, impacting data quality and confidence in downstream conclusions. Automation addresses these challenges directly.
Platforms such as the EggSorter bring AI-powered automation to the earliest and most critical step of toxicity assays: embryo screening, selection, and plating. By standardizing embryo quality criteria and removing operator-dependent variability, automated systems ensure consistent, high-quality sample preparation.
This approach delivers:
- Consistent embryo selection based on defined morphological and developmental parameters
- Improved reproducibility across experiments and operators
- Higher throughput, enabling scalable study designs
- Reduced manual handling, minimizing human bias and fatigue
By automating embryo selection and plating, assays become more standardized from the start, supporting higher-quality data, stronger statistical confidence, and more reliable outcomes.
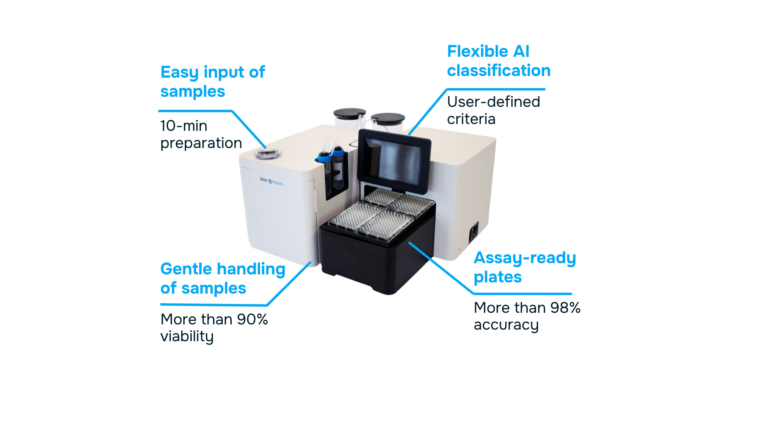
Use Case: The EggSorter & OECD TG 236 (FET)
The OECD TG 236 Fish Embryo Acute Toxicity (FET) test requires fertilized, developmentally synchronized zebrafish embryos confirmed at early cleavage stages (≤ 2 hours post fertilization). Embryos must be morphologically normal prior to exposure.
Although the guideline defines a minimum of 20 embryos per concentration across at least 5 concentrations plus a control, most pre-regulatory and CRO studies operate at a significantly larger scale (6). A common configuration includes 96 embryos per concentration across 5 concentrations and a control. In many high-throughput settings, 3 replicates are included.
OECD TG 236 does not specify a formal timing requirement for embryo selection, as this step is performed manually. In practice, visual inspection under a stereomicroscope typically requires ~3–6 seconds per embryo, with an additional 3–5 seconds for transfer into multi-well plates, resulting in an average handling time of 6–11 seconds per embryo. These estimates are consistent with standard zebrafish husbandry workflows, published facility SOPs, and commonly reported picking rates in embryo manipulation procedures.
In this context, automated platforms such as the EggSorter can streamline embryo screening, selection and plating immediately after collection. Using AI-based image identification, embryos are assessed, selected, and dispensed according to predefined criteria, reducing manual intervention during the most time-sensitive phase of the assay.
The impact becomes clear when comparing manual and automated workflows under a typical high-throughput OECD 236 configuration:
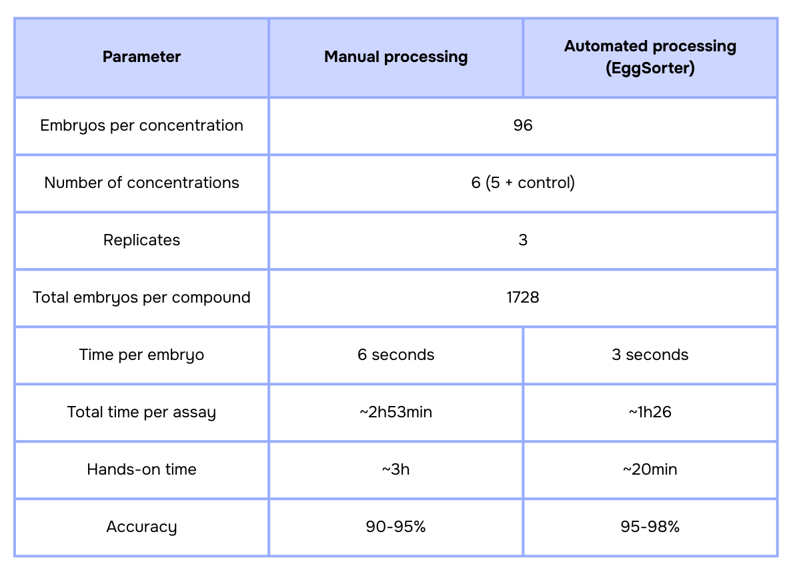
In this scenario, using the EggSorter reduces the total processing time by almost 50% and decreases hands-on time by more than 80%, while standardizing embryo selection criteria across plates.
Regarding reproducibility, even small deviations in selection become statistically relevant at high embryo counts. Manual screening can introduce operator variability, subjective morphology thresholds, fatigue-related inconsistencies, and uneven embryo distribution across plates.
For example, a 2-3% deviation represents 35–50 embryos of potentially different developmental quality. This may shift baseline mortality, increase variability in dose–response curves, and widen dispersion between replicates. As inclusion error rises, mean control mortality can increase from ~5% toward ~7–9%, moving closer to OECD validity limits and increasing the risk of repeat testing.
Because embryo screening and plating occur at the very start of the FET assay, variability introduced at this stage can propagate throughout the study. By reducing operator variability during this critical early phase, automation solutions such as the EggSorter improve reproducibility, increase throughput, and strengthen overall data robustness while maintaining full alignment with OECD TG 236 requirements.
Conclusions & Outlooks
Fish embryo and life-cycle assays provide complementary insights into toxicity, from acute measurement to developmental disruption and reproductive effects. When integrated thoughtfully, these models generate layered information, from early morphological endpoints to generational impacts, strengthening hazard identification and risk assessment.
As regulatory science evolves toward NAMs, combining standardized fish assays with automation and AI-driven embryo selection enhances data quality, reproducibility, and scalability. These synergies support more efficient testing strategies while advancing the reliability and ethical alignment of environmental toxicology.
References
- Santoriello, C., & Zon, L. I. (2012). Hooked! Modeling human disease in zebrafish. The Journal of clinical investigation, 122(7), 2337–2343. https://doi.org/10.1172/JCI60434
- Wang, W., Gao, X., Liu, L., Guo, S., Duan, J.-a., & Xiao, P. (2025). Zebrafish as a vertebrate model for high-throughput drug toxicity screening: Mechanisms, novel techniques, and future perspectives. Journal of Pharmaceutical Analysis, 15(9), Article 101195. https://doi.org/10.1016/j.jpha.2025.101195
- Bambino, K., & Chu, J. (2017). Zebrafish in Toxicology and Environmental Health. Current topics in developmental biology, 124, 331–367. https://doi.org/10.1016/bs.ctdb.2016.10.007
- Zhou, W., Chen, J., Liu, P., Wang, F., & Chen, H. (2024). Comparative effects of different metals on the Japanese medaka embryos and larvae. Ecotoxicology (London, England), 33(6), 653–661. https://doi.org/10.1007/s10646-024-02762-y
- Philippe, C., Grégoir, A. F., Janssens, L., Pinceel, T., De Boeck, G., & Brendonck, L. (2017). Acute and chronic sensitivity to copper of a promising ecotoxicological model species, the annual killifish Nothobranchius furzeri. Ecotoxicology and environmental safety, 144, 26–35. https://doi.org/10.1016/j.ecoenv.2017.05.047
- Busquet, F., Strecker, R., Rawlings, J. M., Belanger, S. E., Braunbeck, T., Carr, G. J., Cenijn, P., Fochtman, P., Gourmelon, A., Hübler, N., Kleensang, A., Knöbel, M., Kussatz, C., Legler, J., Lillicrap, A., Martínez-Jerónimo, F., Polleichtner, C., Rzodeczko, H., Salinas, E., Schneider, K. E., … Halder, M. (2014). OECD validation study to assess intra- and inter-laboratory reproducibility of the zebrafish embryo toxicity test for acute aquatic toxicity testing. Regulatory toxicology and pharmacology : RTP, 69(3), 496–511. https://doi.org/10.1016/j.yrtph.2014.05.018